 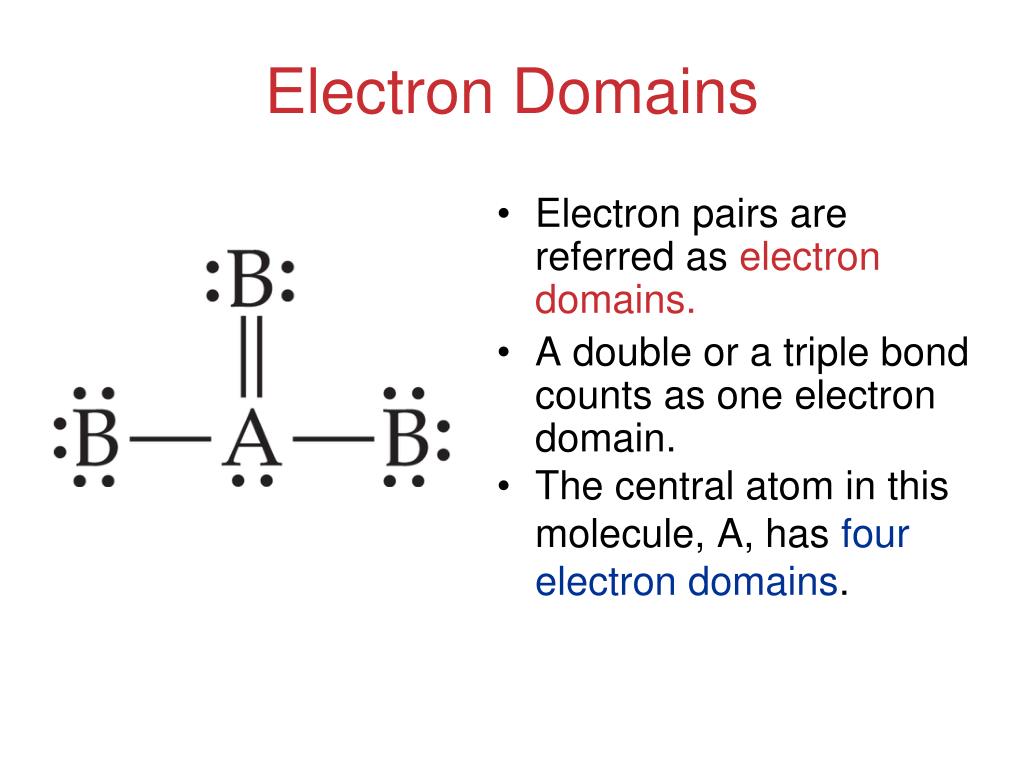 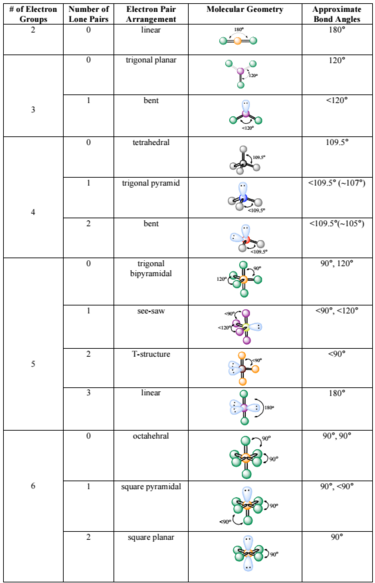
The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms.įundamentally, the VSEPR model theorizes that these regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. At low temperatures, the fluorination reaction is used. VSEPR Theory: a chemistry model used to predict the shape of individual molecules based on electron-pair electrostatic repulsion It is highly unstable and decomposes above the temperature of -28 degrees Celsius.The main geometries without lone pair electrons are: linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral. There are five basic electron domain geometries.There are a total of 4 electron density regions around the central S atom in the Lewis structure of SO32. The sulfur (S) is present at the center of the molecular ion while oxygen (O) occupies the terminals, one on each side. Molecular geometries take into account the number of atoms and the number of lone pair electrons. The Lewis structure of sulfite SO32- ion is made up of a sulfur (S) atom and three oxygen (O) atoms.Choose a central atom: Both the atoms will be central. Look for the total number of bonds forming: Double covalent bonds are forming in an O2 molecule. Molecular Geometry Chart of Electron Groups Number of Lone Pairs Electron Pair Arrangement Molecular Geometry Approximate Bond Angles 2 0 linear 180° 0 trigonal planar 120° 1 3 bent <20° 0 tetrahedral 109.5° 1 trigonal pyramid 4 <109.5° (107°) 2 bent <109. Find how many electrons are needed: It is four for one O2 molecule. The NH 4+ ion is overall non-polar (net µ 0) due to its symmetrical shape and geometry. Find total valence electrons: It is two for each oxygen atom. The NH 4 + ion has an identical electron geometry and molecular geometry or shape i.e., tetrahedral. Fundamentally, the VSEPR model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. The positive 1 charge present on the ion accounts for 1 valence electron removed in its Lewis structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
